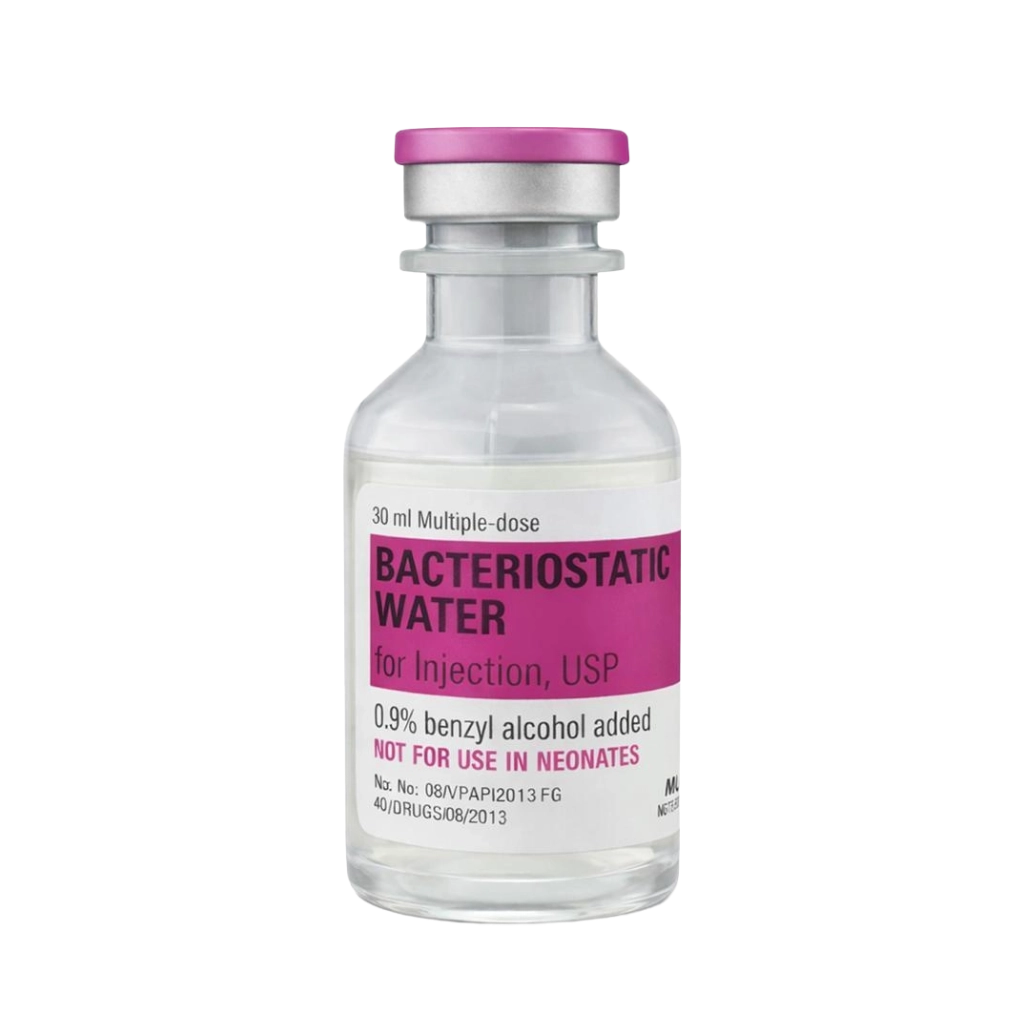
Hospira Bac Water (30ml)
$17.99
Only 5 left in stock
- These products are intended for laboratory research purposes only, and are not for human or animal consumption. The products are not intended to diagnose, treat, cure, or prevent any disease. The products should not be used as a food, drug, cosmetic, or other household use.
Description
Hospira Bacteriostatic Water for Injection is a pharmaceutical-manufactured sterile reconstitution solution produced to USP and cGMP standards. Containing 0.9% benzyl alcohol as a bacteriostatic preservative, it is the highest available purity tier for multi-dose peptide reconstitution in research settings — with defined sterility, non-pyrogenicity, and endotoxin compliance.
Overview
Hospira Bacteriostatic Water for Injection is produced under regulated pharmaceutical manufacturing conditions with defined limits for sterility, endotoxin levels, and particulate matter — providing the highest available assurance of purity for research reconstitution use. The benzyl alcohol component inhibits microbial growth through membrane disruption, enabling safe multi-dose use of reconstituted vials over extended periods.
As a pharmaceutical-grade product with manufacturing traceability, it is the preferred standard in research settings where supply documentation and experimental reproducibility are required.
Key Features
- USP-grade sterile water for injection; manufactured to cGMP pharmaceutical standards
- 0.9% benzyl alcohol preservative for sustained multi-dose antimicrobial protection
- Defined sterility, endotoxin, and particulate matter limits — pharmaceutical manufacturer quality
- Non-pyrogenic; sterile-filtered; clear, colorless solution
- 30mL multi-dose vial format for extended protocol use
- Highest purity tier available for lyophilized peptide reconstitution
Common Research Uses
- Pharmaceutical-grade reconstitution standard for lyophilized peptide research preparations
- cGMP-compliant diluent for protocols requiring documented supply quality
- Multi-dose reconstitution medium for GH secretagogues, metabolic peptides, and regenerative blends
- Reference-standard diluent where manufacturing traceability is required
Product Specifications
- Format: Sterile aqueous solution, USP-grade (0.9% benzyl alcohol); cGMP-manufactured
- Vial size: 30mL multi-dose vial
- Storage: Room temperature (15–30°C) before first use; refrigerate at 2–8°C after opening; discard within 28 days of first puncture
Disclaimer: For research purposes only. Not for human consumption.
RELATED PRODUCTS